|
The deoxy- designation refers to the lack of an alcohol, -OH, group as will be shown in detail further down. Ribose is used in R NA and deoxyribose is used in D NA. It's only when it's in the bonded acetal form that it's locked into either the alpha or beta configuration. Ribose and its related compound, deoxyribose, are the building blocks of the backbone chains in nucleic acids, better known as DNA and RNA. Keep in mind that the hemiacetal form can interconvert with the ring-open carbonyl form, and the "anomeric position" of the monosaccharide is free to invert in chirality - that is, it can readily flip between "up" and "down". (One thing that might be tripping you up is the directionality of the 1 carbon with respect to the 2 carbon. 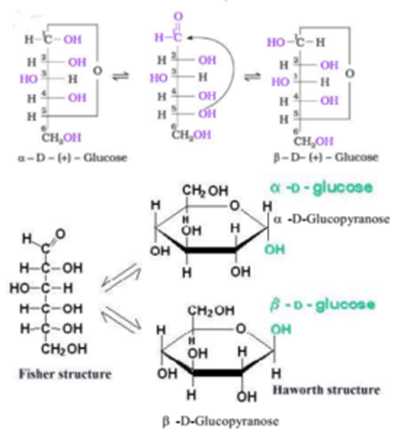
Both of these carbons are numbered "2", and if you imagine rotating the furanose ring 180 degrees to flip over that acetal position, you'll see the other carbons match up as well. You have the ring oxygen of the furanose, and then a free hydroxyl oxygen. Therefore, some copper(II) complexes were prepared. In the second picture, it's the carbon on the far right. from publication: Investigations of carbohydrate isomers and metal. In the first image, this is the carbon which is making the oxygen bridge between the two monomers (the one on the left of the furanose) - the oxygen of the bridge is one oxygen, and the oxygen of the furanose ring is the other. The cyclic form of glucose is called glucopyranose. That is, you're looking for a carbon with multiple bonds to oxygens. The pyranose ring is formed by attack of the hydroxyl on carbon 5 of glucose to the aldehyde carbon (carbon 1, also called the anomeric carbon in carbohydrate terminology). Instead of looking for an aldehyde or a ketone in the cyclic form, you're looking for an acetal or hemiacetal. When you have a linear Fischer projection (like the "D-fructose (linear)" images on the second image) the carbonyl carbon should be obvious. Are you looking for an answer to the topic Where are the anomeric carbons in lactose We answer all your questions at the website in category: +15 Marketing Blog Post Ideas And Topics For You.You will find the answer right below. The rest of the carbons are numbered accordingly. For ketoses, it's the lowest number that can be reached in the linear, ring-open form (e.g. For aldoses, this carbon would be carbon 1. The configuration, and hence the contribution by C 2 to C 5 part remains same for both the anomers. For a given aldohexoses, the contribution by the two configurations at C 1 carbon, the anomeric carbon atom, are regarded as +A and A. 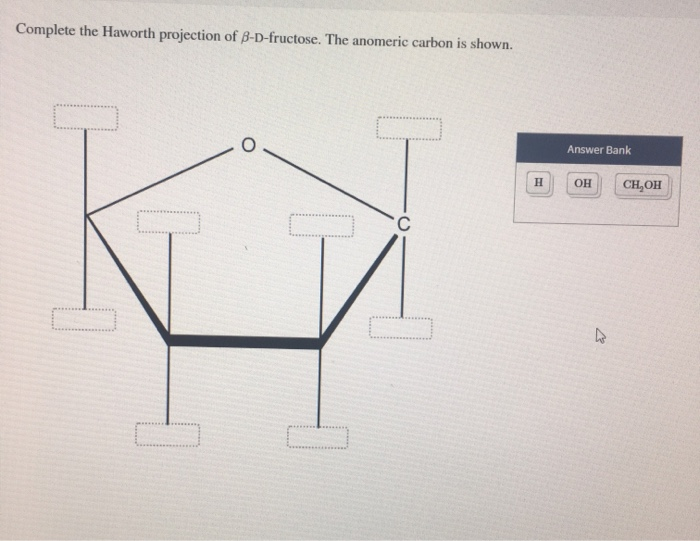
This will be the end of the monosaccharide which is numbered lowest. Fructose is a ketohexose that exists as a crystalline powder with a density of 1.55 g/mL. The key point in carbohydrate numbering is to look for the carbon which is an aldehyde or a ketone (the carbonyl carbon). Actually, the numbering is the same, at least for the fructose portion of the molecule (which is the right-hand monosaccharide in your sucrose image - the one on the left is glucose).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
